ABSTRACT
Data have been accumulating on the risk of developing type 2 diabetes in patients receiving statins and on the potential adverse effects of these drugs on glycemic control in patients who already have type 2 diabetes. This article reviews data linking statin use and new-onset diabetes mellitus, the effects of statins on glycemic control in type 2 diabetes, the benefit-risk considerations of statin use and type 2 diabetes, and how these factors affect patient management.
The weight of the evidence suggests that statin use is associated with an increased risk of new-onset diabetes mellitus, but the magnitude of the effect has varied across studies, including differences between randomized controlled trials and observational studies.
The risk is generally greater with high-intensity statin therapy and higher statin doses. However, many other variables are also involved, including prediabetes, genetics, central obesity, dyslipidemia, hypertension, lifestyle, and other medications, most notably glucocorticoids.
In patients with type 2 diabetes mellitus, statin use is associated with a small increase in hemoglobin A1c, and this effect may be greater with atorvastatin than with other statins. However, the benefits of statins in preventing atherosclerotic cardiovascular disease outweigh their adverse effects on glycemic control.
Pitavastatin has been associated with a lower risk of new-onset diabetes mellitus, but it lacks data on cardiovascular outcomes from large trials in patients with diabetes.
After the first of the statins was introduced in 1987, a number of clinical trials demonstrated that these drugs, which effectively lower low-density lipoprotein cholesterol (LDL-C) levels, consistently reduce the risk of atherosclerotic cardiovascular disease events in many types of patients, including those with type 2 diabetes mellitus.1 One of these trials—the West of Scotland Coronary Prevention Study (WOSCOPS),2 with 5,974 patients—even reported that statins decreased the risk of new-onset diabetes.
Then, a larger trial—Justification for the Use of Statins in Prevention: an Intervention Trial Evaluating Rosuvastatin (JUPITER)3 with 17,802 participants treated with rosuvastatin or placebo—found a higher incidence of diabetes in the rosuvastatin group.
This observation raised questions and stimulated efforts to find answers. Do statins cause diabetes? If they do, is it true for all statins? Is the risk a function of the potency and dose of the statin? Are there other risk factors for this effect? In patients who already have diabetes, do statins worsen glycemic control? Most importantly, do the benefits of statin use in reducing cardiovascular risk outweigh any increased risk of new-onset diabetes mellitus?
Two caveats are warranted. First, in most studies, new-onset diabetes was diagnosed by the individual investigators and not according to any protocol. It was probably underdiagnosed, but in double-blind trials, the amount of underdiagnosis was likely about the same in each treatment group. Second, there are not enough data on microvascular complications of diabetes mellitus in statin-treated patients to warrant any discussion of risk vs benefit. The available results are limited and inconsistent regarding statin benefit or harm for microvascular complications, and they vary for retinopathy, nephropathy, and neuropathy.
DO STATINS CAUSE DIABETES? A NEW LOOK AT OLD TRIALS
Investigators have examined data from a number of trials of statin therapy in preventing cardiovascular disease events to look for effects on the incidence of new-onset diabetes mellitus.
The JUPITER trial3 reported the incidence of physician-reported new-onset diabetes mellitus during the 1.9-year trial duration as 3.0% in the rosuvastatin group and 2.4% in the placebo group (P < .01).
Rajpathak et al4 performed a meta-analysis of 6 trials, including JUPITER3 and WOSCOPS.2 The risk of new-onset diabetes mellitus was increased by 6% when WOSCOPS was included (P = NS) and by 13% (P= .008) when WOSCOPS was excluded.4
Sattar et al5 performed another meta-analysis of the same 6 trials plus 7 more, for a total of 13. The results varied widely across trials (Table 1).5 In 4 trials, the incidence of new-onset diabetes was higher in the control group than in the statin group, while it was higher in the statin group in the other 9.5 Individually, none of the findings was statistically significant, but combined, the odds ratio for new-onset diabetes was 1.09 with statins, which was statistically significant (95% confidence interval [CI] 1.02–1.17). The investigators calculated that 255 patients would need to be treated with statins for 4 years to observe 1 extra case of diabetes.5
Statins and diabetes: Results of 13 trials
The Women’s Health Initiative6 recruited 161,808 postmenopausal women without diabetes mellitus at baseline, of whom 153,840 had enough data to be analyzed post hoc. Statin therapy was associated with a 71% higher risk of new-onset diabetes mellitus (self-reported). After adjustment for age, body mass index, family history of diabetes, and other variables, the risk was still 48% higher in statin users.6
Lin et al7 used a database of more than 30,000 patients who had undergone percutaneous intervention for acute coronary syndromes. Propensity score matching (n = 9,043 in each group) was used to evaluate the effects of statin use vs no statin use on new-onset diabetes. In the unmatched cohort, statin use was actually associated with a lower risk of diabetes. However, in the matched cohort the risk of new-onset diabetes was higher in statin users than in nonusers (adjusted hazard ratio 1.27, 95% CI 1.14– 1.41, P ≤ .001). The hazard ratios varied depending on which statin the patients received, and they were all statistically significant except for lovastatin (and the risk was lower in lovastatin users than in those not receiving any statin at all). Hazard ratios were as follows:
Lovastatin 0.87
Atorvastatin 1.30
Fluvastatin 1.38
Rosuvastatin 1.42
Pravastatin 1.71.7
Engeda et al8 performed a meta-analysis of 8 randomized controlled trials and 15 observational studies. They found an association between statin use and new-onset diabetes mellitus and also showed that the risk was higher in observational studies (relative risk 1.55, 95% CI 1.39–1.74) than in randomized controlled trials (relative risk 1.11, 95% CI 1.00–1.22).8
In summary, the weight of the evidence suggests that statin use is associated with an increased risk of new-onset diabetes mellitus, but the magnitude of the effect varied across studies, including differences between randomized controlled trials and observational studies.
DO ALL STATINS DO IT? DOES RISK VARY BY STATIN INTENSITY?
Statin potency, now more commonly called statin intensity, is based on the amount by which each statin lowers LDL-C and at what dose. Statins are thus classified as high-, moderate-, or low-intensity (Table 2),9 and this classification is widely used in guidelines for giving statins to prevent atherosclerotic cardiovascular disease based on the patient’s baseline risk of cardiovascular events.9
Categories of statin therapy
Dose-response relationships among statins and their effects on LDL-C levels have been well known since the first statins came on the market.10 Since proprotein convertase subtilisin/kexin type 9 inhibitors, which lower LDL-C levels even more than statins do, have not been reported to increase the risks of dysglycemia or new-onset diabetes, any relationships between statins and diabetes risk is likely related to the statin itself and not to the LDL-C reduction.11
A number of studies have addressed whether statin dose and intensity are related to the risk of new-onset diabetes mellitus.
Preiss et al12 reported on 5 large clinical trials comparing statins in higher vs lower doses and showed that each of them had an odds ratio point estimate greater than 1 for higher doses to increase incident diabetes. The odds ratio for the pooled estimate was 1.12 (95% CI 1.04–1.22).12
Other investigators have analyzed higher- and lower-intensity statins, each compared with placebo.
Sattar et al,5 in the meta-analysis described above, calculated that the overall odds ratios for incident diabetes were nominally higher with rosuvastatin (1.18), atorvastatin (1.14), and simvastatin (1.11) than with pravastatin (1.03) and lovastatin (0.98); recall that for all 13 studies combined it was 1.09 (Table 1).5 The analysis could not adjust for all confounders such as age, new-onset diabetes mellitus being more common in older patients.5
Navarese et al13 performed a network meta-analysis of 17 randomized controlled trials (14 placebo-controlled and 3 that compared 2 doses of statins) with a total of 113,394 participants. The data generally suggested a relationship between statin intensity as well as higher vs lower doses of some statins and risk of diabetes. The incidence of new diabetes was highest with rosuvastatin and lowest with pravastatin.
Carter et al14 performed a population-based study using information from several databases in Ontario, Canada. Data from 471,250 patients who did not have diabetes at baseline and were treated with a statin supported the idea that the risk was greater with rosuvastatin, atorvastatin, and simvastatin than with lovastatin and fluvastatin in both primary and secondary cardiovascular disease prevention cohorts. The incidence of diabetes was also a function of duration of exposure.
In summary, the weight of the evidence from randomized controlled trials and observational studies suggests that higher-intensity statins and higher doses of statins are associated with higher risk of new-onset diabetes mellitus.
RISK FACTORS FOR DIABETES IN NON–STATIN-TREATED PATIENTS
Phenotypic and laboratory variables that may be associated with risk of new-onset diabetes mellitus in general include the following:
Fasting blood glucose levels
Postprandial glucose levels
Triglyceride levels
Hypertriglyceridemia, including elevated ratio of triglyceride to high-density lipoprotein cholesterol
Hypertension
Elevated measures of obesity, including body mass index and waist-to-hip ratio
Smoking
Depression
Hyperuricemia
Sleep disturbances
Gestational diabetes
Polycystic ovary disease.
RISK FACTORS FOR DIABETES IN STATIN-TREATED PATIENTS
Many of the same risk factors associated with the development of diabetes in the general population are also present in statin users who develop associated diabetes.17–20
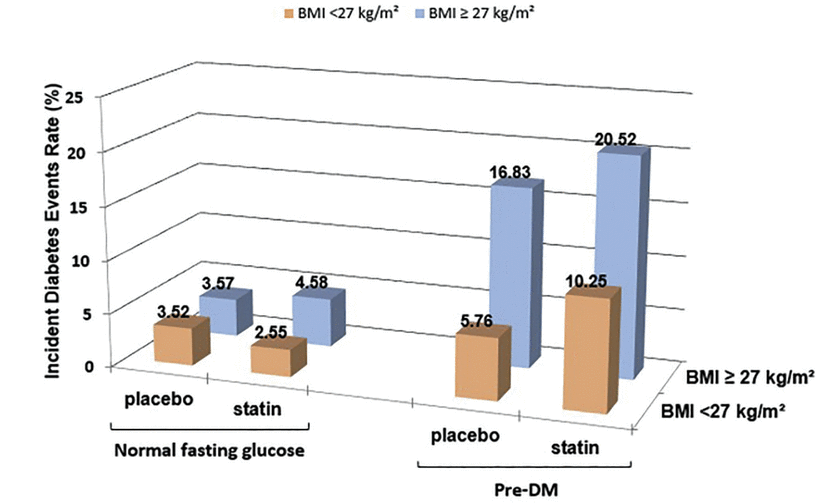
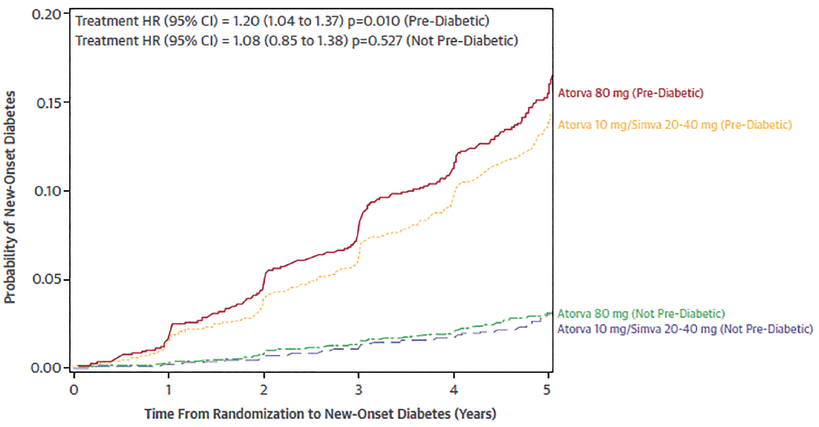
Kohli et al,21,22 in an analysis of 2 large trials of atorvastatin,19,20 reported that the risk of new-onset diabetes in patients who had high triglyceride levels or high body mass index depended on whether they had prediabetes, ie, fasting blood glucose or hemoglobin A1c levels higher than normal but not high enough to be classified as diabetes. Statins had little effect on the incidence of diabetes in those who did not have prediabetes (Figure 1, Figure 2).21 The risk continued to diverge over time between the group with prediabetes and the group without prediabetes, and was greatest in those with prediabetes receiving atorvastatin 80 mg daily (Figure 3).21
Risk of new-onset diabetes according to statin use, prediabetes, and elevated triglyceride (TG) level in the Treating to New Targets trial and the Stroke Prevention by Aggressive Reduction in Cholesterol Levels trial. For triglycerides, 1.7 mmol/L = 150 mg/dL.
Reprinted from Am J Cardiol, Vol 118(9), Kohli P, Knowles JW, Sarraju A, Waters DD, Reaven G. Metabolic markers to predict incident diabetes mellitus in statin-treated patients (from the Treating to New Targets and the Stroke Prevention by Aggressive Reduction in Cholesterol Levels Trials), pages 1275–1281; 2016, with permission from Elsevier.
Risk of new-onset diabetes by statin use, prediabetes, and body mass index (BMI) in the Treating to New Targets trial and the Stroke Prevention by Aggressive Reduction in Cholesterol Levels trial.
Reprinted from Am J Cardiol, Vol 118(9), Kohli P, Knowles JW, Sarraju A, Waters DD, Reaven G. Metabolic markers to predict incident diabetes mellitus in statin-treated patients (from the Treating to New Targets and the Stroke Prevention by Aggressive Reduction in Cholesterol Levels Trials), pages 1275–1281; 2016, with permission from Elsevier.
Risk of new-onset diabetes by statin use, prediabetes, and body mass index in the Treating to New Targets and the Stroke Prevention by Aggressive Reduction in Cholesterol Levels trials (Atorva = atorvastatin; CI = confidence interval; HR = hazard ratio).
Reprinted from Am J Cardiol, Vol 118(9), Kohli P, Knowles JW, Sarraju A, Waters DD, Reaven G. Metabolic markers to predict incident diabetes mellitus in statin-treated patients (from the Treating to New Targets and the Stroke Prevention by Aggressive Reduction in Cholesterol Levels Trials), pages 1275–1281; 2016, with permission from Elsevier.
Among the 15,056 participants who did not have diabetes at baseline, 5,924 (39%) had a fasting glucose level between 100 mg and 126 mg (designated prediabetes) and 9,132 (61%) had normal fasting glucose levels.22 Statin treatment was balanced between groups. As in other studies, the participants with prediabetes were older and more likely to have features of metabolic syndrome, hypertension, higher triglyceride levels, and lower HDL-C levels. During a mean 5-year follow-up, 14.2% of participants with prediabetes developed new diabetes compared with 2.9% of those without prediabetes.22
High-intensity statin use in prediabetes was associated with a higher risk of new diabetes, but in patients with normal fasting glucose levels there was no difference in diabetes risk between those who received high-intensity vs low-intensity statins.22
Arsenault et al23 assessed 18 biomarkers associated with the risk of cardiovascular disease in the Treat to New Targets trial and found that plasma lipids, adiponectin, and lipoprotein-associated phospholipase A2 might be also useful for predicting incident diabetes in statin-treated patients.
In summary, variables associated with increased risk of progression to diabetes in general cohorts are also associated with an increased risk of diabetes in statin-treated patients. The statin-associated risk of diabetes is most evident in patients with high fasting blood glucose levels and prediabetes. These observations suggest that statin use is an additional risk factor for new-onset diabetes.
DO STATINS WORSEN GLYCEMIC CONTROL?
Statin use was associated with increasing hemoglobin A1c and fasting blood glucose levels in patients with established type 2 diabetes in some studies.24–31 Several small randomized controlled trials had designs that allowed analyses of statin effects on glycemic control.
Simsek et al24 reported that hemoglobin A1c values increased by 0.3% with both atorvastatin 80 mg/day and rosuvastatin 40 mg/day over 18 weeks.
The AFORRD trial (Atorvastatin in Factorial With Omega-3 EE90 Risk Reduction in Diabetes),25 in 800 patients with type 2 diabetes, reported that hemoglobin A1c increased by the same amount (0.3%) with atorvastatin 10 mg over 4 months.
Teramoto,26 in contrast, found that hemoglobin A1c decreased with pitavastatin.
Three meta-analyses addressed the effects of statins on glycemic control in type 2 diabetes.
Zhou et al27 analyzed 26 statin trials that included 3,232 participants and concluded that statin therapy “had no remarkable influence” on hemoglobin A1c. The mean change was 0.04%.27
Erqou et al28 performed a meta-analysis of 9 placebo-controlled trials of atorvastatin, simvastatin, and pravastatin. Overall, the hemoglobin A1c levels were 0.12% higher in the statin groups than in the control groups after an average follow-up of 3.6 years, with most of the effect related to atorvastatin use. This analysis is confounded by inadequate information on any changes in use of glucose-lowering agents.
Cui et al29 performed a network meta-analysis of 23 trials to assess the overall effects of statin and statin dosage on hemoglobin A1c. Overall, in statin users, hemoglobin A1c increased by 0.11%. The greatest effect was with high-intensity atorvastatin, which was associated with a mean increase of 0.63%. Pitavastatin was associated with a reduction in hemoglobin A1c compared with all other statins.
The Fremantle Diabetes Study,30 an observational study in Australia, found that low-intensity statins (n = 119) were not associated with any change in hemoglobin A1c, moderate-intensity statins (n = 195) were associated with a mean increase of 0.22% (P = .022), and high-intensity statins (n = 11) were associated with a mean increase of 1.05% (P = .023).
Sukhija et al31 reported changes in fasting blood glucose in a large database of US Veterans Affairs patients with type 2 diabetes mellitus. In unadjusted analyses, fasting blood glucose increased from 102 mg/dL to 141 mg/dL in statin users and from 100 mg/dL to 129 mg/dL in statin nonusers.
In summary, any conclusions about the effects of statins on glycemic control, especially hemoglobin A1c, in type 2 diabetes are confounded by the type of analyses, the limited data on any changes in glucose-lowering agents, and study durations and size. Overall, the data support the conclusion that there is a small aggregate effect of statins in increasing hemoglobin A1c. This effect may be greatest with high-dose atorvastatin and least with pitavastatin.
HOW MIGHT STATINS INCREASE DIABETES RISK?
Historically, type 2 diabetes has been largely characterized by 2 major metabolic abnormalities: insulin resistance and declining beta-cell function. However, many other abnormalities including inflammation, glucagon dysregulation, and altered renal thresholds for glycosuria are also associated with it.
Mechanistic studies in animals and humans have sought links between statin use and diabetes risk. One of the more likely possible explanations is statin-associated insulin resistance.32–35 This hypothesis has not been tested in the large studies of new-onset diabetes mellitus or studies of changes in hemoglobin A1c in type 2 diabetes mellitus.
BENEFIT OUTWEIGHS RISK
Diabetes has been consistently shown to be associated with an increased risk for cardiovascular disease, and many clinical trials have shown that statin treatment is associated with a reduction in cardiovascular disease risk.1 Most patients with type 2 diabetes mellitus are at high risk of atherosclerotic cardiovascular disease because they also have other risk factors such as obesity, hypertension, and the high triglyceride-to-low HDL-C ratio often associated with insulin resistance. Thus, the obvious concern revolves around whether the risk of developing diabetes with statin alters the benefit-risk considerations and whether statin use significantly attenuates the benefits of statins on atherosclerotic disease risk and events.
Ridker et al36 analyzed the JUPITER3 data and concluded, “the cardiovascular and mortality benefits of statin therapy exceed the diabetes hazard, including in participants at high risk of developing diabetes.”36 The benefit-risk question has also been addressed in thoughtful reports by Navarese et al37 and Collins et al.38 Each of these groups concluded that the evidence supports the contention that in patients with a high risk for atherosclerotic disease, the benefits of statin use outweigh the risk of new-onset diabetes. Navarese et al noted that the benefit-risk considerations are less clear for patients without diabetes and 0 to 1 risk factors.37 Collins et al stated that “the absolute benefits of statin therapy depend on the individual’s absolute risk of occlusive vascular events and the absolute reduction in LDL cholesterol that is achieved.”38
GUIDELINES SUGGEST SHARED DECISION-MAKING
The 2018 guidelines9 suggest that in patients at low risk of atherosclerotic cardiovascular disease based on a low 10-year risk score and no diabetes, starting a statin is recommended only after discussing it with the patient. This approach aligns with the considerations raised by Navarese et al.37
If patients have a calculated 10-year risk of 7.5% or higher but no other significant risk factors, besides elevated LDL-C, the guidelines suggest a discussion about starting a moderate-intensity statin, eg, pravastatin 40 mg, lovastatin 40 mg, fluvastatin 80 mg, or pitavastatin 1 to 4 mg daily. Each of these is associated with a lower risk of new-onset diabetes mellitus than the high-intensity statins.
Some data suggest that pitavastatin actually lowers the risk of diabetes, but this drug lacks the extensive cardiovascular disease outcomes data in patients with diabetes that exist for the other statins,1 and this fact may be a point of discussion between the healthcare team and patient.
If patients have a calculated 10-year risk of 7.5% to 19.9%, especially when associated with additional risk factors, then the guidelines favor starting an intermediate-intensity statin. As noted above, the risk of new diabetes with statin use is higher in people who already have multiple atherosclerotic cardiovascular disease risk factors.9,37
In patients without type 2 diabetes and at low 10-year risk of atherosclerotic cardiovascular disease, if statin use is agreed on between the healthcare team and patient, a low-intensity statin is likely the best consideration.
In patients without type 2 diabetes but with multiple risk factors and a high risk for atherosclerotic cardiovascular disease over 10 years, the use of moderate- or high-intensity statins is justified, as the benefits of statins outweigh the risk of developing diabetes.
The guidelines do not discuss how statin therapy may change in a patient who develops new-onset diabetes mellitus related to statin use, but it has been addressed by Collins et al.38 In cardiovascular disease risk calculators, diabetes is a yes-or-no question, and having diabetes approximately doubles one’s risk. However, this may be overly simplistic, as the relationship between glucose and cardiovascular risk is continuous and graded.39 In general, benefits of both moderate-intensity and high-intensity statins on reducing the risk for atherosclerotic cardiovascular disease events outweigh risks associated with hyperglycemia.
In patients with type 2 diabetes who are treated with moderate- or high-intensity statins, careful follow-up of hemoglobin A1c and appropriate glucose-lowering therapy should be implemented. Glucose-lowering therapies with established benefits on atherosclerotic cardiovascular disease are preferred.
Backes et al40 have distilled these recommendations into a digestible format, which I have combined with those of the guidelines in Table 3.9,40,41
Starting statin therapy: Things to consider and discuss
DISCLOSURES
Dr. Hoogwerf has disclosed ownership interest in Eli Lilly and consulting for Mannkind and Zealand Pharmaceuticals.
- Copyright © 2023 The Cleveland Clinic Foundation. All Rights Reserved.