ABSTRACT
Treatment of resistant hypertension is a challenge, especially in patients who have chronic kidney disease. The choice of medications may be limited in this group, making the possibility of device-based therapies attractive. We explore 4 devices and procedures available to treat this vexing issue.
Placing a shunt between the iliac artery and iliac vein (arteriovenous coupling) relieves the arterial pressure hemodyamically; this approach is experimental.
Catheter ablation of sympathetic nerve endings in the renal artery leads to less activation of the renin-angiotensin-aldosterone system and lower blood pressure; this procedure is experimental as well.
A third experimental approach to lowering blood pressure is stimulation of the carotid baroreceptors with an implanted pacemaker or stent device.
For patients with renal artery stenosis, percutaneous re-vascularization with stent placement can be considered; the current American College of Cardiology/American Heart Association guidelines give the procedure a class IIa recommendation.
Nonpharmacologic, device-based anti-hypertensive treatments show potential, but, except for stenting of the renal arteries in patients who have renal artery stenosis, all remain experimental. Researchers have focused on patients with chronic kidney disease (CKD) and resistant hypertension, a group at high risk, in whom the benefit may justify the risk and cost of the treatment.
See related editorial, page 444
Nonpharmacologic procedure-based treatments that could in theory provide a permanent cure would be welcome in this group of patients. We discuss the role of several procedure-based treatments, ie, arteriovenous coupling, renal sympathetic denervation, baroreflex activation, and renal percutaneous revascularization in the management of resistant hypertension (Table 1).
Advantages and limitations of antihypertensive procedures
RESISTANT HYPERTENSION IS COMMON IN CKD
Resistant hypertension is defined as blood pressure that remains above goal despite concurrent use of 3 antihypertensive agents of different classes (1 of which is a diuretic) at their maximum tolerated doses, or controlled blood pressure with the use of 4 or more agents.1
The prevalence of true resistant hypertension is difficult to ascertain, and patients suspected of having it should undergo a meticulous search for reversible causes (Table 2).
Possible causes of difficult-to-treat hypertension
CKD, defined by the presence of kidney damage or decreased kidney function for 3 or more months irrespective of the cause, presents a unique challenge in patients with resistant hypertension.2–4 The prevalence of apparent treatment-resistant hypertension in this group is estimated to range from 23% to 42%, and it is associated with worse prognosis.5–7 Maintaining normal blood pressure can be difficult, given features that are common in CKD such as accelerated atherosclerosis, fluctuating volume status, inability to use the full spectrum of antihypertensive medications due to increased adverse effects, and related nonadherence issues.8 The task is made more challenging by the revised hypertension guidelines,2 which encourage clinicians to target blood pressure below 130/80 mm Hg in patients with CKD.
ARTERIOVENOUS COUPLING
Peripheral arteriovenous fistulae created for hemodialysis access are known to reduce vascular resistance. Based on this principle, there have been efforts to add a low-resistance, high-compliance venous tract parallel to the high-pressure systemic arterial circulation to reduce arterial resistance and pressure.9
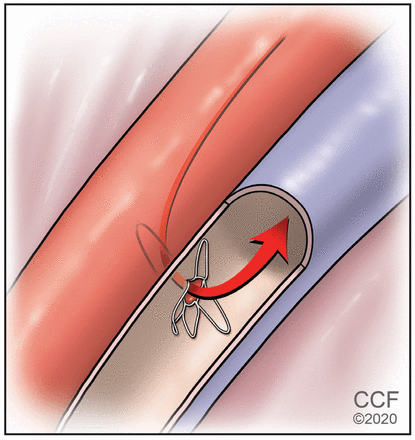
The ROX coupler (ROX Medical, San Clemente, CA) is a device placed between the distal iliac vein and artery to create a central arteriovenous anastomosis (Figure 1).
A percutaneously placed device creates an arteriovenous anastomosis.
Trial of arteriovenous coupling
In an initial trial,10 44 patients were randomly assigned to receive the device and 39 were assigned to receive normal care. Six months later, office systolic blood pressure had dropped by a mean of 26.9 (standard deviation 23.9) mm Hg in the device group (P < .0001) and by 3.7 (21.2) mm Hg in the control group (P = .31).
There was no deterioration in renal function at 6 months, though patients with advanced CKD (estimated glomerular filtration rate [eGFR] < 30 mL/min/1.73 m2) were excluded from the study.10 However, the trial lacked a sham treatment control group, treatment adherence was not verified, and proximal venous stenosis developed in nearly 29% of the intervention group, tempering the positive results.11
A subsequent trial was planned that would have included a control group undergoing a sham procedure , but it seems to have been terminated by the sponsor.12
Prospects for arteriovenous coupling
It remains unclear at this stage if arteriovenous coupling has the potential to become a mainstream procedure.
RENAL SYMPATHETIC DENERVATION
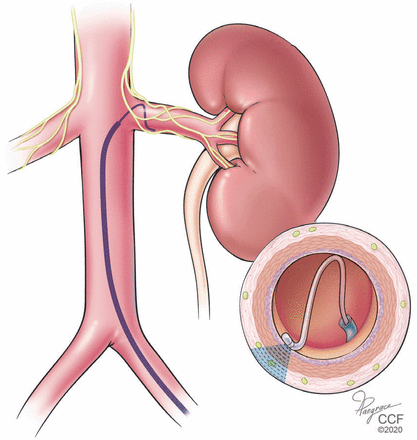
Hyperactivity of the sympathetic nervous system is known to be a major factor in sustaining resistant hypertension. Curtailing this hyperactivity to better control blood pressure is a potential treatment for resistant hypertension.13,14 With this view, catheter-based devices have been developed to ablate the sympathetic afferent and efferent nerves of the renal artery by radiofrequency or ultrasound energy (Figure 2) or by transarterial injection of caustic substances.15
Renal artery denervation is performed using an intra-arterial catheter.
Trials of renal sympathetic denervation
Despite success in early studies, subsequent trials have had discouraging results.16 Pooled data from 3 sham-procedure-controlled trials of first-generation devices showed no significant reduction in blood pressure on summary treatment estimates (weighted mean difference 2.23 mm Hg, 95% confidence interval [CI] –4.70 to 0.25 mm Hg; P = .08).17
Most of these trials excluded patients with eGFRs lower than 45 mL/min/1.73 m2, but even so, renal denervation did not seem to have a major deleterious effect on renal function. In the non-sham-controlled SYMPATHY trial (N = 139), the average eGFR was 77 ± 19 mL/min/1.73 m2 at baseline and declined by 1.5 (−3.1 to 0.1) mL/min/1.73 m2 at 6 months, with no difference between the groups receiving renal denervation and usual medical care.18,19
A subsequent sham-controlled trial set out to include participants with low eGFR, but only 3% of the denervation treatment group had eGFRs between 30 and 45 mL/min/1.73 m2; most (92%) had eGFRs higher than 60. There was no detectable change in renal function after the procedure.20
In recent sham-controlled trials, novel second-generation devices seemed to hold promise. In the SPYRAL HTN-ON MED and OFF-MED trials,21,22 renal denervation using the Symplicity Spyral device (Medtronic, Dublin, Ireland) led to statistically significant and clinically meaningful blood pressure reduction at 6 months; the mean 24-hour systolic blood pressure had dropped by 7.0 mm Hg (95% CI –12.0 to –2.1; P = .0059). Patients with eGFR less than 45 mL/min/1.73 m2 were excluded, but no patient in the entire cohort had a serum creatinine elevation greater than 50%, and no new or worsening renal failure was reported.
The RADIANCE-HTN SOLO trial23 showed that renal denervation with the Paradise system (ReCor Medical, Palo Alto, CA) reduced daytime ambulatory systolic blood pressure in 74 patients (8.5 vs 2.2 mm Hg; P < .0001). While patients with eGFR less than 40 were not recruited, there were no significant changes in eGFR between the treatment groups (adjusted mean difference –0.6, 95% CI –4.4 to 3.2, P = .75).
Cost-effectiveness of renal denervation
Geisler et al24 calculate that the discounted lifetime incremental cost-effectiveness ratio for renal denervation therapy is $3,071 per quality-adjusted life-year, and the 95% credible interval for incremental cost-effectiveness ratio is $31,460 per quality-adjusted life-year.
Chowdhury et al25 report that over a lifetime at the current estimated costs, renal denervation it would be cost-effective only if it were targeted to patients whose 10-year predicted cardiovascular risk was at least 13.2% initially.
Prospects for renal denervation
Improvement in the design of renal denervation delivery could overcome some of the procedural setbacks of earlier trials. This, and better selection of patients, may lead to acceptable results of renal denervation in the near future. Experience suggests that patients who have mild CKD may tolerate this treatment well. However, well-designed, adequately powered trials to evaluate the long-term efficacy and safety of second-generation renal denervation technology in patients with resistant hypertension with all stages of CKD are needed to validate the safety of this treatment in CKD.
BAROREFLEX ACTIVATION THERAPY
Another approach to reducing sympathetic tone to help control blood pressure is electric stimulation of the carotid sinus barore-ceptors.26 The first-generation Rheos system (CVRx, Minneapolis, MN) consisted of a pacemaker unit implanted subcutaneously in the infraclavicular position along with electrodes leading to both carotids.27
Studies of baroreflex activation therapy
Early studies using the Rheos system in 383 patients showed that substantial blood pressure reduction was maintained over a follow-up of 6 years.28 Patients on dialysis were generally excluded from these trials, and those with CKD made up only a small portion of the cohort (< 15%).
In a follow-up study of 236 patients from the Rheos Pivotal trial, the mean eGFR decreased from 92 ± 20 mL/min at baseline to 87 ± 22 mL/min at 6 months in the active therapy group and to 85 ± 23 mL/min in the control group (P = .589). Given the drop in eGFR in both groups, this decrease could merely represent the normal decline of renal function over time. In the relatively small subgroup of patients with an eGFR less than 60 mL/min (n = 18, mean eGFR 49 ± 8 mL/min), renal function remained stable over a 12-month observation period.29
The second-generation Neo system (also from CVRx), which uses a smaller electrode, was developed to mitigate some of the procedure-related complications such as cranial nerve injuries associated with the use of first-generation Rheos.
In a pilot study in 23 patients with CKD and resistant hypertension who were treated with the second-generation Neo system, there was a significant decrease in the mean arterial blood pressure (116.9 ± 20.9 mm Hg before vs 104.2 ± 22.2 mm Hg after the procedure). Patients who had stage 3 or 4 CKD experienced a greater reduction in proteinuria, and the eGFR remained stable in the treated patients despite the reduction of systemic blood pressure.30
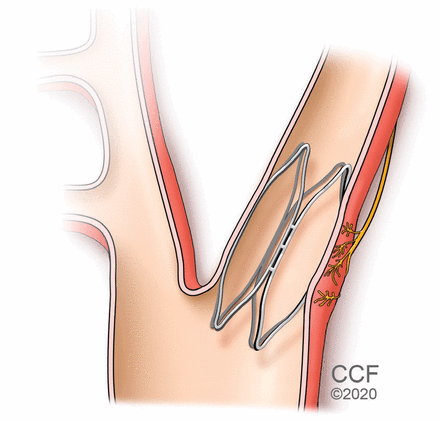
The MobiusHD device (Vascular Dynamics, Mountain View, CA), another second-generation device, is a catheter-delivered self-expanding intracarotid implant designed to activate the baroreflex (Figure 3).31 In its first study in humans, it seemed successful in reducing blood pressure, and a larger trial designed to evaluate the safety and effectiveness of the MobiusHD device is actively enrolling patients.31,32 Again the long-term efficacy and safety of second-generation baroreflex activation devices in patients with resistant hypertension and CKD of all stages is yet to be verified in large randomized controlled trials.
A percutaneously placed implant is designed to stimulate the carotid barore-ceptors and thus lower blood pressure.
Cost-effectiveness of baroreceptor stimulation
Borisenko et al33 calculate that baroreceptor stimulation therapy generates 1.66 additional life-years and 2.17 additional quality-adjusted life years at an incremental cost of €16,891 compared with continued medical management in a simulated cohort of 50-year-old patients at high risk of end-organ damage. Baroreceptor stimulation was estimated to be cost-effective compared with optimal medical treatment with an incremental cost-effectiveness ratio of €7,797 per quality-adjusted life year.
However, an independent assessment by the Norwegian Institute of Public Health noted that based on incremental cost-effectiveness ratio levels and after adjusting the model to account for important shortcomings in the submitted analysis related to clinical effect and health-related quality of life, the incremental cost-effectiveness ratio rises well above the level that has been considered cost-effective in Norway.34
RENAL ARTERY STENTING
Renal artery stenosis compromises blood flow to the kidneys, activating the renin-angiotensin-aldosterone axis and causing hypertension. In more than 90% of cases, renal artery steno-sis is due to atherosclerosis, usually affecting the ostial part of the renal artery.35
Clinicians are encouraged to suspect renal artery stenosis and to look for it in patients with resistant hypertension, as it has been noted to be present in up to 24% of these patients.36 Risk factors and specific clinical presentations that raise suspicion for renal artery stenosis are presented in Table 3 and Table 4.
Causes of renal artery stenosis
Clues to the presence of renal artery stenosis
Optimal medical therapy remains the preferred treatment of atherosclerotic renal artery stenosis. Major society guidelines emphasize optimal medical therapy with blockade of the renin-angiotensin-aldosterone axis to confer survival benefit in these patients.37
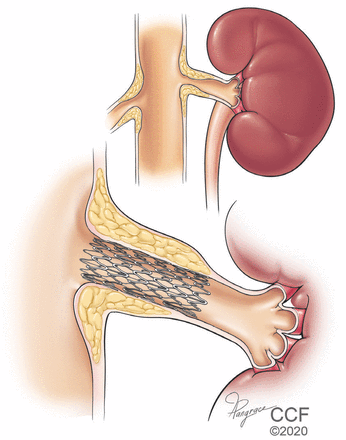
However, clinicians and researchers have long hoped that procedural intervention could relieve renal artery stenosis, cure the hypertension, and eliminate the burden of lifelong medical therapy. Pioneering work by Grüntzig et al38 with balloon angioplasty of renal artery stenosis showed significant relief of hypertension. The subsequent development of vascular stents led to percutaneous revascularization by stenting as the preferred technique to resolve renal artery stenosis (Figure 4).39
Stenting is reasonable for treating resistant hypertension in patients with significant renal artery stenosis.
Early case series and registries seemed to validate the utility of percutaneous resvascularization as a treatment for renal artery stenosis. In a nonrandomized single-arm study of 202 patients (with 241 total lesions), percutaneous resvascularization lowered the mean systolic blood pressure from 162 mm Hg at baseline to 145 mm Hg at 9 months (P < .0001), while the eGFR remained nearly the same at 58 vs 57 mL/min/1.73 m2 (P = .38).40
However, these results could not be replicated in various subsequent randomized controlled trials.41 Analysis of 8 trials, which included 2,223 patients, showed that renal artery revascularization was not associated with a change in systolic blood pressure from baseline when compared with medical therapy (weighted mean difference 0.12, 95% CI −0.97 to 1.21, P = .83). Moreover, revascularization was not associated with a reduced incidence of adverse cardiovascular or renal outcomes, and the results seemed similar when restricted to 5 stent-only trials.41
However, the randomized controlled trials may not tell the whole story. Design flaws, patient selection, and enrollment bias in various published trials may limit their clinical applicability, especially in patients who might benefit the most.42
There hence seems to be a broad expert consensus that certain groups of patients with severe renal artery stenosis should be treated with revascularization. The current American College of Cardiology/American Heart Association guidelines on the management of peripheral arterial disease give the procedure a class IIa recommendation (level of evidence B), stating that percutaneous revascularization “is reasonable” for patients with hemodynamically significant renal artery stenosis and resistant hypertension.37,43 Similarly, a Society for Cardiovascular Angiography and Interventions statement also suggests percutaneous revascularization may be considered as appropriate care in patients with significant renal artery stenosis and resistant hypertension.44
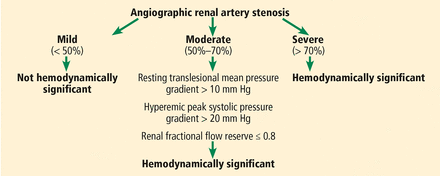
Figure 5 presents diagnostic criteria for significant renal artery stenosis and outlines when percutaneous revascularization can be considered.
Hemodynamic significance of angiographic renal artery stenosis.
Renal outcomes after percutaneous revascularization have varied. In one of the largest randomized controlled trials to date, the Cardiovascular Outcomes in Renal Atherosclerotic Lesions (CORAL study), with 947 patients in total, the rates of end points were similar between the percutaneous revascularization group and the medical therapy-only group at 43 months of follow-up, including death from renal causes (2 cases vs 1, P = .6), progressive renal failure (77 vs 89, P = .34), and need for permanent renal replacement therapy (16 vs 8, P = .11).45
In the Angioplasty and Stenting for Renal Artery Lesions (ASTRAL) trial,46 with 806 randomized patients, the rate of progression of renal impairment was slightly slower in the revascularization group than in the medical group (−0.07 × 10−3 L/μmol/year vs −0.13 × 10−3 L/μmol/year; P = .06) over 5 years of follow-up. Over the same time, the mean serum creatinine level was 1.6 μmol/L lower in the revascularization group than in the medical therapy group.
Thus, percutaneous revascularization for renal artery stenosis seems to have a reasonable renal safety profile even in patients with CKD.
Cost-effectiveness of percutaneous revascularization
In a German study47 analyzing the cost-effectiveness of medical therapy, percutaneous transluminal angioplasty with and without a stent, and surgery for the therapy of renal-artery stenoses in hypertensive patients, the average reimbursed treatment cost per patient after 3 years was as follows:
€9,121 for medication
€17,164 for surgery
€14,670 for percutaneous angioplasty
€8,437 for stenting.
This resulted in cost-effectiveness ratios per event-free patient at 3 years as follows:
€51,752 for medical treatment
€36,454 for surgery
€78,766 for percutaneous angioplasty
€11,663 for stenting.
The researchers concluded that a strategy of primary stent implantation is more cost-effective than stand-alone balloon dilatation.47
THESE TREATMENTS MAY PROVE USEFUL
Nonpharmacologic treatments for resistant and difficult-to-treat hypertension in patients with CKD may prove to be useful. Percutaneous revascularization may be considered in patients with resistant hypertension and underlying renal artery stenosis.
Ongoing trials have demonstrated the efficacy and safety of newer renal denervation and baroreflex activation devices, but more data are needed regarding treating the difficult subgroup of hypertensive patients who have CKD of all stages. The concept of reducing pill burden and increasing medication adherence remains attractive and has a large potential for improving outcomes in this high-risk group.
We would like to emphasize that except for renal artery stenting, the therapies discussed here remain experimental and are not approved by the US Food and Drug Administration for routine clinical use except as part of clinical trials.
Footnotes
Dr. Rader has disclosed consulting for ReCor Medical.
- Copyright © 2020 The Cleveland Clinic Foundation. All Rights Reserved.