ABSTRACT
COVID-19 is primarily considered a respiratory illness, but the kidney may be one of the targets of SARS-CoV-2 infection, since the virus enters cells through the angiotensin-converting enzyme 2 receptor, which is found in abundance in the kidney. Information on kidney involvement in COVID-19 is limited but is evolving rapidly. This article discusses the pathogenesis of acute kidney injury (AKI) in COVID-19, its optimal management, and the impact of COVID-19 on patients with chronic kidney disease, patients with end-stage kidney disease on dialysis, and kidney transplant recipients.
AKI is common in COVID-19 and is associated with poor outcomes.
SARS-CoV-2 can damage the kidney through several mechanisms, including acute lung injury, sepsis, hemodynamic alterations, cytotoxic effects, cytokine release syndrome, rhabdomyolysis, coagulopathy, microangiopathy, and collapsing glomerulopathy.
Despite initial speculation, renin-angiotensin-aldosterone system inhibitors need not be discontinued in patients with COVID-19.
Treatment of AKI includes general management, pharmacologic management of COVID-19, hemodynamic and volume optimization, and extracorporeal therapies.
Pharmacotherapy for COVID-19 can be divided into antibacterial, antiviral, immunomodulatory, and anti-inflammatory drugs.
Severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2), the cause of coronavirus disease 2019 (COVID-19), gains entry into target cells through the angiotensin-converting enzyme 2 (ACE2) receptors. ACE2 receptors are present in the kidneys as well as the lungs, heart, and intestinal cells.1–8
The renin-angiotensin system plays an important role in human physiology. Angiotensin I is cleaved from angiotensinogen by renin and converted to angiotensin II by ACE. Angiotensin II causes systemic vasoconstriction and also enhances inflammation, endothelial cell dysfunction, oxidative stress, collagen synthesis in fibroblasts, and fibrosis in target organs.7,9 ACE2 is a counterregulatory enzyme that breaks down angiotensin II to form angiotensin 1–7, which mediates vasodilation and attenuates angiotensin II-mediated inflammation.7
SARS-CoV, the virus that caused the SARS epidemic in 2003, downregulates expression of ACE2 after it enters the cell, and without the counterregulatory effects of ACE2, the deleterious effects of angiotensin II are believed to lead to lung disease, including severe acute respiratory distress syndrome.7,10
The kidney has an abundance of ACE2 receptors and therefore may be one of the primary targets of SARS-CoV-2 infection.7 ACE2 is expressed in the kidney much more than in the lungs, specifically on the brush border apical membrane of the proximal tubule and also at lower levels in the podocytes.11 Virus particles were observed in the tubular epithelium and in podocytes in an autopsy series of COVID-19 patients.12 TMPRSS2 is robustly expressed in the distal nephron but not the proximal tubule; it is unclear if other transmembrane serine proteases in the proximal tubule can mediate the priming step.8
INCIDENCE RATES VARY IN DIFFERENT REPORTS
The reported incidence of acute kidney injury (AKI) in COVID-19 ranged from 0.5% to 56.9% in various case series (Table 1).4–6,13–22 The wide range is likely related to different definitions used and different populations studied.
COVID-19: Incidence of acute kidney injury and need for kidney replacement therapy
A higher incidence has been reported in the United States than in China.4–6,13–19 Also, studies from China reported the onset of AKI within a median of 7 to 14 days after admission, whereas a large study of patients hospitalized with COVID-19 in the United States17 found that the onset tended to be early; 1,993 (36.6%) of 5,449 US patients developed AKI, and of these, 37.3% either arrived with it or developed it within 24 hours of admission. Of those with AKI, 31.1% reached stage 3 (the highest, defined as an increase of 3 times or more in serum creatinine within 7 days or start of kidney replacement therapy), and 14.3% needed kidney replacement therapy.
In another study,20 79 (31%) of 257 critically ill patients required kidney replacement therapy. A study contrasting AKI incidence between 3,345 patients with COVID-19 and 1,265 patients without COVID-19 hospitalized during the same time period showed that those with COVID-19 had a higher incidence of AKI (56.9% vs 37.2%), and more of them needed kidney replacement therapy (4.9% vs 1.6%).19
Black race has been shown to be independently associated with a higher risk of AKI in COVID-19.17,19
Higher mortality rate
AKI in COVID-19 is also associated with a higher risk of death.13,21 A systematic review and meta-analysis23 of 6 studies from China found that severe AKI in COVID-19 (defined as AKI stage 3 and AKI requiring kidney replacement therapy) was associated with a 3-fold higher risk of death.
A US study17 reported a mortality rate of 35% in patients with AKI; of those who died, 91% had stage 3 AKI. The mortality rate was 55% in those needing kidney replacement therapy. Another study19 showed an in-hospital mortality rate of 33.7% in those with COVID-19-associated AKI compared with 13.4% in those with AKI without COVID-19. Those with stage 3 AKI and COVID-19 had a 2.6-fold higher mortality rate than those with stage 3 AKI who did not have COVID-19.
There are limited data on the long-term prognosis of COVID-19 patients with AKI. A single-center study from China reported that although the mortality rate was high in patients with AKI, nearly half of the patients recovered from AKI within 3 weeks of onset of infection.21 A US study17 reported a median creatinine level of 1.70 mg/dL (interquartile range 0.96, 3.50) at the time of discharge in patients with AKI, and 91% of hospitalized patients who needed kidney replacement therapy were still on it at the time of study censoring. Another study from the United States showed that fewer COVID-19 AKI patients recovered renal function than AKI patients without COVID-19 (42.3% vs 68.5%).19
Proteinuria and hematuria have been reported in patients with COVID-19, but their significance and impact on mortality are not yet known. An early report from China13 found proteinuria in 43.9% and hematuria in 26.7% of patients. Subsequent studies from the United States reported 2+ or 3+ blood in 46% of patients, and 2+ or 3+ protein in 42% of patients on urine dipstick analysis. Another study showed proteinuria in 87% of patients.17,20
While the effects of ACE2 expression and viral entry on proteinuria are unknown, it is speculated that proteinuria could be related to viral replication, particularly in the podocytes.11
PATHOGENESIS OF ACUTE KIDNEY INJURY
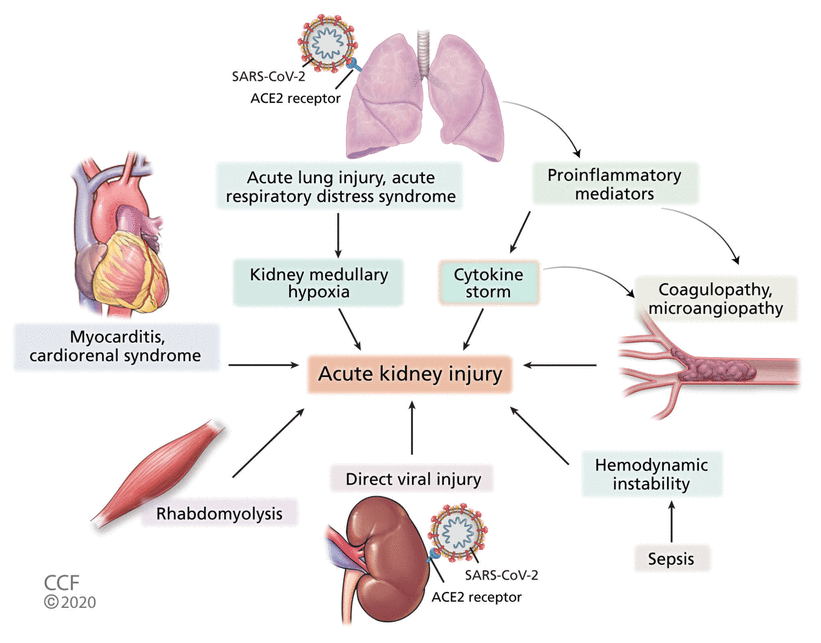
Figure 1 shows the possible pathophysiologic mechanisms of AKI in patients with COV-ID-19.
Pathophysiology of acute kidney injury in COVID-19 (ACE2 = angiotensin-converting enzyme 2; SARS-CoV-2 = severe acute respiratory syndrome coronavirus 2).
Acute lung injury
Acute lung injury could lead to AKI through hemodynamic changes and reduced cardiac output with high intrathoracic pressure, inflammatory cytokines that lead to systemic inflammation, and reduction in kidney medullary perfusion due to hypoxemia.24–26
Sepsis
Sepsis, due to viral or bacterial infection, is associated with increased risk of AKI. Higher levels of inflammatory cytokines including interleukin 6 (IL-6), and maladaptive immune responses leading to microvascular dysfunction, increased vascular permeability, and tissue damage have been suggested as mechanisms of AKI in sepsis, along with hypoperfusion affecting the kidney microcirculation.27
A study from China early in the pandemic reported septic shock in 6.4% of COVID-19 patients.4 Another study found that 59% of COVID-19 patients developed sepsis, 20% developed septic shock, and 15% developed secondary infection.15 Bacterial co-infections have been reported in severely ill COVID-19 patients, which raises the possibility of sepsis playing a role in AKI in these patients.16
Hemodynamic alterations
Cardiorenal syndrome is another possible mechanism of AKI in patients with COVID-19. Viral myocarditis and cardiomyopathy with left ventricular dysfunction could lead to decreased perfusion to the kidneys, resulting in AKI.26
Cytotoxic effects leading to tubular and podocyte injury
SARS-CoV-2 can potentially injure tubular cells and podocytes, leading to proteinuria, hematuria, and AKI. ACE2 is expressed in high amounts in the proximal tubular cells and podocytes, which may be the site of viral entry in the kidneys,28 and studies have noted proteinuria and hematuria in COVID-19 patients.13,17,20
In an autopsy series by Su et al,12 light microscopy showed proximal acute tubular injury with occasional frank tubular necrosis. Corona virus-like particles were also observed under electron microscopy in the proximal tubular epithelium and podocytes.12,29
In a preprint report of autopsy data on 6 patients from China,30 viral nucleoprotein antigens were detected in the kidney tubules by immunohistochemical analysis, and virus-like particles were observed under electron microscopy. However, the rabbit monoclonal antibody against the SARS-CoV-2 nucleoprotein used in the autopsy series to detect virus particles has been described as having nonspecific positive staining of the kidney parenchyma in non-COVID-19 patients.31
Larsen et al31 reported that in situ hybridization for SARS-CoV-2 did not detect viral RNA in the kidney.
Thus, there is conflicting evidence regarding the possible direct cytopathic effects of SARS-CoV-2 on tubular epithelial cells.
Cytokine release syndrome or hyperinflammation
Viral and bacterial infections are known to cause excessive release of inflammatory cytokines that lead to organ damage.32 Huang et al33 reported that in the 2003 outbreak, SARS-CoV infection resulted in cytokine storm or hyperinflammation with increased levels of IL-6, IL-8, and interferon gamma, which led to increased vascular permeability and diffuse alveolar damage. As described above, patients with acute lung injury have an increased risk of AKI due to hemodynamic changes, hypoxia, and inflammatory cytokines.
Rhabdomyolysis
Case reports from the 2003 SARS epidemic described rhabdomyolysis of suspected viral etiology and AKI, but evidence of causation has been lacking.34,35 Postmortem kidney histopathologic analysis of COVID-19 patients has shown pigmented casts in the kidney tubules and increased creatine kinase, possibly representing rhabdomyolysis of unclear etiology.12
Coagulopathy and microangiopathy
Coagulopathy has been noted in COVID-19 patients, with altered prothrombin time, activated partial thromboplastin time, D-dimer levels, fibrinogen levels, and fibrin degradation product levels and disseminated intravascular coagulation.36,37 Release of inflammatory mediators and the uninhibited effects of angiotensin II can possibly trigger the coagulation cascade and predispose to hypercoagulability.38 A high incidence of thrombotic complications has been reported even in those on prophylactic anticoagulation.15,39–41 In a postmortem kidney histopathologic analysis, fibrin thrombi were present in the absence of red blood cell fragmentation and platelet thrombi, due to which a hypercoagulable state was suspected.12
Thus, endothelial cell dysfunction leading to activation of the coagulation cascade and thrombosis of the microcirculation may also play a role in AKI.42
Collapsing glomerulopathy
Collapsing glomerulopathy has been reported in kidney biopsies of patients with COVID-19.31,43,44 In one report,44 tubuloreticular inclusions were observed, which can be associated with viral infections. It is hypothesized that either the direct viral effect or presence of increased cytokines from the systemic inflammatory response, or both, can lead to a collapsing variant of focal segmental glomerulosclerosis, especially in patients with high-risk alleles of the APOL1 gene.43
MANAGEMENT OF ACUTE KIDNEY INJURY
Management of patients with a confirmed diagnosis of COVID-19 and AKI begins with an evaluation of the cause of AKI. A broad framework of prerenal, renal, and postrenal causes should be considered.
Taking the history, performing a physical examination, and ascertaining the timeline of AKI through chart review are important for diagnosis and management. Medications should be carefully reviewed, and any potentially nephrotoxic agents should be discontinued if possible. A Foley catheter should be considered to accurately measure urine output and to relieve possible obstruction.
Blood work, urinalysis, urine sediment examination for cells and casts, and imaging may help diagnose different causes of AKI.45 Table 2 summarizes laboratory and imaging studies that can help differentiate the causes.
Causes of acute kidney injury in COVID-19 patients
The treatment of AKI can be divided into general management, pharmacologic management of COVID-19, optimizing hemodynamic and volume status, and extracorporeal therapies. Vaccines against COVID-19 are currently under development, but none has been licensed for use so far.46
Renin-angiotensin system inhibitors and COVID-19
Initial concerns were raised regarding a possible association of renin-angiotensin system inhibitors (including ACE inhibitors and angiotensin II receptor blockers) with increased risk of COVID-19, due to possible increased expression of ACE2 based on animal models, and hence increased possibility of viral entry. However, there is no clinical evidence of harm with renin-angiotensin system inhibitors in COVID-19.47–51
In an observational analysis,50 renin-angiotensin system inhibitors were not associated with increased risk of testing positive for COVID-19 or developing severe COVID-19. Likewise, in a population-based case-control study in Italy,49 there was no evidence of increased risk of COVID-19 with the use of ACE inhibitors or angiotensin II receptor blockers.
Renin-angiotensin system inhibitors are also thought to have a possible protective effect through decreasing the level of angiotensin II, which is thought to exert an inflammatory effect.7,52 Therefore, these drugs should not be discontinued in stable patients with COVID-19, and this has been emphasized by professional societies including the American College of Cardiology, American Heart Association, and European Society of Hypertension.53,54 For inpatients with COVID-19, the decision to discontinue renin-angiotensin system inhibitors should be based on hemodynamic and clinical status, as well as kidney function trend.52
Pharmacologic management of COVID-19
Pharmacologic management of COVID-19 can be divided into antibacterial therapy, antiviral therapy, and immunomodulatory and anti-inflammatory therapy. Broad-spectrum antibiotics are usually started to treat secondary bacterial infection.55
Clinical trials are ongoing, and there are no drugs currently approved by the US Food and Drug Administration (FDA) for the treatment of COVID-19. However, the FDA issued emergency use authorizations for remdesivir and convalescent plasma for hospitalized patients with COVID-19, and recently broadened the scope of remdesivir to include all hospitalized patients with COVID-19 regardless of severity.56–58
A full review of pharmacologic therapy for COVID-19 is beyond the scope of this review article, but Table 3 lists selected drugs, their mechanisms of action, current evidence, and possible nephrotoxic effects.7,56–72
Selected therapies for COVID-19 a
Optimizing hemodynamic and volume status
Hemodynamic alterations due to infection, and acute respiratory distress syndrome leading to impaired gas exchange, hypoxia, and right heart failure, can increase the risk of AKI. COVID-19 may lead to cardiogenic shock due to myocarditis and the resultant decrease in cardiac output, or distributive shock due to cytokine release syndrome, sepsis-induced systemic vasodilation, or both. Cytokine release may also lead to hypovolemic shock due to capillary leak and loss of intravascular volume.73,74
Therefore, optimizing hemodynamic and volume status is important. Early volume resuscitation should be initiated in hemodynamically unstable patients to reverse hypoperfusion to vital organs, particularly when patients first present with evidence of volume depletion due to fever and respiratory distress. A fine balance needs to be achieved, avoiding overly aggressive fluid resuscitation and fluid overload on the one hand, and overly conservative resuscitation and hypoperfusion on the other, with frequent hemodynamic assessment and echocardiograms. Therefore, an active approach with early resuscitation and early termination of fluid resuscitation should be implemented.75
Vasopressor therapy is needed to support blood pressure in patients with shock.
Diuretic therapy should be considered if volume overload is suspected in a hemodynamically stable patient, as in cardiorenal syndrome. Volume overload refractory to diuresis requires ultrafiltration, a form of kidney replacement therapy.
Extracorporeal organ support
Treatment of multiorgan dysfunction in critically ill patients with COVID-19 may necessitate extracorporeal organ support, including kidney replacement therapy, extracorporeal membrane oxygenation, and a left ventricular assist device.26
Kidney replacement therapy
Up to 31% of critically ill patients with COVID-19 require kidney replacement therapy for severe AKI.4–6,14–16,18,20 The indications for it in COVID-19 patients with AKI are the same as for other AKI patients. There is no evidence at this time to suggest a benefit for starting it early vs later.
Hemodynamically unstable patients on vasopressor therapy are started on continuous kidney replacement therapy and later transitioned to intermittent hemodialysis or peritoneal dialysis when their hemodynamic status improves. Convective forms of dialysis (hemofiltration) may in theory remove cytokines better but have not been proven to lead to better outcomes than diffusive forms. Continuous ultrafiltration can help reverse hypervolemia in cardiorenal syndrome and increased intra-abdominal pressure that may contribute to abdominal compartment syndrome.26
Challenges in kidney replacement therapy
A major challenge during continuous kidney replacement therapy in COVID-19 patients is frequent circuit clotting, thought to be due to upregulation of the coagulation system by inflammatory cytokines.76 We have observed that circuit clotting seems to improve with heparin use, particularly when administered through the circuit (prefilter).
Also, malposition of the dialysis catheter tip should be ruled out. A short catheter length with the tip in the superior vena cava can lead to slow flows and catheter dysfunction.77 Figure 2 shows the chest radiograph of a patient with COVID-19 who had a short catheter that clotted frequently, and the optimal location of a tunneled dialysis catheter to optimize blood flow, which is essential in patients with increased clotting.
(Top) Radiography shows a short dialysis catheter with its tip in the superior vena cava in a patient with COVID -19 with frequent clotting. (Bottom) Illustration of the optimal location of the dialysis catheter in the mid-atrium.
Hemodialysis in patients with acute respiratory distress syndrome who require prone positioning needs a coordinated and sequential timing protocol to provide adequate ventilatory support in the prone position and dialysis therapy in the supine position. A synchronized team approach should be implemented to coordinate and maintain the safety of vascular access during prone positioning. Prolonged intermittent renal replacement therapy may need to be considered rather than continuous therapy.
To limit exposure of dialysis nurses and technicians, we use extension tubing that allows dialysis machines to be placed outside the patient room. However, extension tubing increases circuit length and thus carries a risk of hypothermia, thrombosis, and blood loss. Therefore, appropriate warming systems should be implemented.78 We have also installed video monitors that allow nurses to observe patients on dialysis without entering the room.
Dialysis, a finite resource
Hospital dialysis units may experience a shortage of dialysis supplies and dialysis technicians and nurses. A careful daily assessment of available dialysis resources is needed: a dialysis dashboard to track equipment, supplies, personnel, and patients should be implemented.
Permissive underdialysis, with shorter dialysis treatments and less-frequent treatments, may be needed to preserve supplies and to allow for treatment of more patients. Strategies to overcome shortages in prepared continuous kidney replacement therapy dialysis fluid includes preparing replacement fluid by inpatient pharmacies.78 Our institution has been producing dialysis machine-generated ultrapure dialysate that was shown to be safe, effective, and economical in a cohort of 405 patients on continuous kidney replacement therapy.79
Other options include using peritoneal dialysis fluid as replacement fluid, or possibly emergently starting peritoneal dialysis using bedside tunneled peritoneal dialysis catheter placement. Nontunneled acute peritoneal dialysis catheter placement, a procedure rarely done in the United States, may be an option in dire situations, but is limited by operator experience and training, and thus only considered as a last resort when all other measures fail.78,80
Extracorporeal membrane oxygenation, direct hemoperfusion, and left ventricular assist devices
Lung and cardiac injury with COVID-19 can lead to hypoxia and decreased kidney perfusion, which can lead in turn to kidney medullary hypoxia and cardiorenal syndrome. Supporting the heart and lung in these conditions using left ventricular assist devices and extracorporeal membrane oxygenation can potentially help with renal perfusion. Direct hemoperfusion using a macroporous sorbent has been suggested as a treatment to adsorb and remove circulating cytokines and prevent cytokine release syndrome-induced end-organ damage.26 All these modalities can be used in conjunction with continuous kidney replacement therapy to help manage the multiorgan failure commonly seen in critically ill patients with COVID-19.26
SPECIAL CONSIDERATIONS
COVID-19 and chronic kidney disease
Many patients with chronic kidney disease have multiple comorbidities such as diabetes and hypertension, which can predispose them to COVID-19. Chronic kidney disease is associated with a higher risk of severe infection.13,81 A meta-analysis81 showed that about 20% of patients with chronic kidney disease who contracted COVID-19 had severe disease, a 3-fold higher risk compared with those without chronic kidney disease. Those with COVID-19 and AKI had a higher prevalence of chronic kidney disease than those without AKI.19
Telemedicine can be used to monitor and manage patients with stable chronic kidney disease while minimizing their exposure.82 The platforms must comply with Health Insurance Portability and Accountability Act standards.
COVID-19 and outpatient maintenance dialysis in end-stage kidney disease
Patients with end-stage kidney disease on maintenance dialysis usually have multiple comorbidities and are at increased risk of COVID-19. Unavoidable patient gathering and frequent travel to outpatient dialysis units can increase their risk of infection. Therefore, preventive strategies should be implemented to minimize transmission and protect patients on outpatient hemodialysis and peritoneal dialysis.83
The US Centers for Disease Control and Prevention, American Society of Nephrology, and International Society for Peritoneal Dialysis have issued interim infection control measures to help mitigate the risk of infection in dialysis patients.84–86 Educating patients and healthcare workers on COVID-19 is imperative.84–86
Peritoneal dialysis patients should keep at least 2 weeks of dialysis supplies on hand. Nonessential visits to dialysis units should be avoided.84 Screening for COVID-19 symptoms, temperature checks, and testing protocols for patients and staff at dialysis units should be implemented. Specific dialysis units can be dedicated to COVID-19 patients. Home hemodialysis, where feasible, is an effective alternative for outpatient hemodialysis that implements social distancing while minimizing the need for frequent travel and transportation.83
COVID-19 and kidney transplant recipients
Kidney transplant recipients are at increased risk of infection, particularly from a depressed T-cell immune response due to immunosuppression.87 The risk is highest during the first 3 months after transplant, particularly if patients receive induction therapy with lymphocyte-depleting agents.88 Therefore, during the COVID-19 pandemic, elective kidney transplant should be performed with caution.88
Screening of donors is imperative because of a high tropism of the virus for the kidney, and programs are currently moving toward testing both donors and recipients before transplant.89 In addition, symptomatic and high-risk donors under clinical suspicion for COVID-19 should postpone donation even if they test negative for SARS-CoV-2, given the potential for false-negative results.89
The evidence on COVID-19 infections in kidney transplant recipients is limited to case reports and case series.87–93 Fever, cough, and myalgias are the most frequently reported symptoms,90 although they are not always present.91 Guillen et al91 described a patient who presented with vomiting, dehydration, and conjunctivitis. In a cohort of 36 kidney transplant recipients, Akalin et al93 reported diarrhea as the second most common symptom, after fever. Moreover, mild symptoms such as low-grade fever, mild cough, and normal white blood cell count have been suggested to occur in kidney transplant recipients due to the protective effect of immunosuppressive therapy against the cytokine storm.92 Therefore, even mild or atypical symptoms should prompt COVID-19 testing in kidney transplant recipients.
The optimal management for kidney transplant recipients with COVID-19 is still being studied. The general consensus is to taper down immunosuppression while simultaneously protecting graft function and initiating antiviral, antibiotic, and anti-inflammatory therapy.87–93 Immunosuppressive therapy should be tapered down to help mount an antiviral immune response and decrease the severity of symptoms.87 Most authors suggest discontinuing antiproliferative drugs such as mycophenolate mofetil, but maintaining calcineurin inhibitor therapy at lower levels along with glucocorticoid therapy.87–93 The role of pulse steroid therapy is controversial, but some authors suggest it has a beneficial anti-inflammatory effect while minimizing graft rejection.87
Antiviral therapy, such as the protease inhibitors ritonavir and lopinavir, was reported to be effective in some patients.89 A possible role for remdesivir has been reported.60 Due to strong drug interactions of protease inhibitors with calcineurin inhibitor therapy, calcineurin inhibitor dosage must be reduced substantially.89,91 Tocilizumab blocks IL-6 receptors and can possibly reduce inflammation.61,94
Most of the patients in the reported cases recovered and their immunosuppressive therapy was gradually reintroduced.87,88,90–92,95
SCIENCE IS EVOLVING RAPIDLY
The science on COVID-19 is rapidly evolving, and new evidence is published on a daily basis. This review is based on our experience and is limited to evidence that is currently available.
AKI in patients with COVID-19 is associated with increased mortality. The etiology is multifactorial, and management is supportive, with possible need for extracorporeal therapies for critically ill patients.
Large-scale prospective clinical trials can help inform optimal management of AKI in COVID-19, and more retrospective data on clinical experience is needed to assess the impact and prognosis of COVID-19 on patients with chronic kidney disease or end-stage kidney disease, and in kidney transplant recipients.
- Copyright © 2020 The Cleveland Clinic Foundation. All Rights Reserved.